Excitement and fear around AI and cognitive computing has reached fever pitch, but are cancer treatment and diagnosis where the technology will have its biggest and most positive impact yet?
“We don’t know what cancer really looks like,” says Helmy Eltoukhy, an electrical engineer from California and the founder of Guardant Health, the first company to commercialise a comprehensive genomic liquid biopsy. “We think we do, but in reality, we are using very simplistic generalised features to categorise cancer.”
Cancer is thought to be a disease of the genome. It took 13 years and three billion dollars before, in 2003, the technology to sequence the first human genome was invented. Since then, the price has fallen – it now costs $1,000 and can be done in one day.
Reaching this milestone will help unlock the secrets of cancer. However, using the large and complex data sets that sequencing produces to achieve better patient treatment and diagnosis is problematic and demanding. So far, the healthcare sector has only scratched the surface – but this is changing thanks to the power of artificial intelligence (AI) and machine learning.
Many tech entrepreneurs, alongside the big players Alphabet’s Verily, Google’s DeepMind and IBM Watson, are adopting cognitive computing to build tools to better understand cancer and other diseases. According to CBI Insights, deals to healthcare-focused AI start-ups increased from fewer than 20 in 2012 to nearly 70 in 2016.
Eltoukhy founded Guardant Health in 2013 to change this. Two years later his company launched the Guardant360 blood test to market.
Used for Stage Three and Stage Four cancer patients, the test unravels genetic sequences of a patient’s cancer to categorise its sub-type without the need for a physical biopsy, which can be dangerous and expensive. Once the mutation that caused the disease is identified, better and more targeted treatment options can be found.
To create the test, it was necessary to unlock the weak genomic alterations released into the blood from cancer that normal genomic sequencing technology is not powerful enough to detect.
To identify these signals, Eltoukhy and his team borrowed concepts from digital communication. He turned to work by his former Stanford University professor, John Trophy, who invented the digital subscriber line (DSL), which enables 100 times faster speed with the same copper phone lines that dial-up modems use.
“The trick there was pre-coding the information that is sent on copper phone lines and decoding on the backend – essentially making information more robust and resistant to noise being introduced on the phone or on the bandwidth property of the phone line,” explains Eltoukhy.
“The challenge in DNA sequencing is instead of putting 0s and 1s in a ‘dry’ form into a copper phone line or communication channel, we are putting four wet letter ACGT molecules into the DNA sequencer,” says Eltoukhy. “They are drying out and the challenge was translating a lot of the concepts of digital communication theory into biology, using the same algorithm to lower the error rate by over a thousand-fold.”
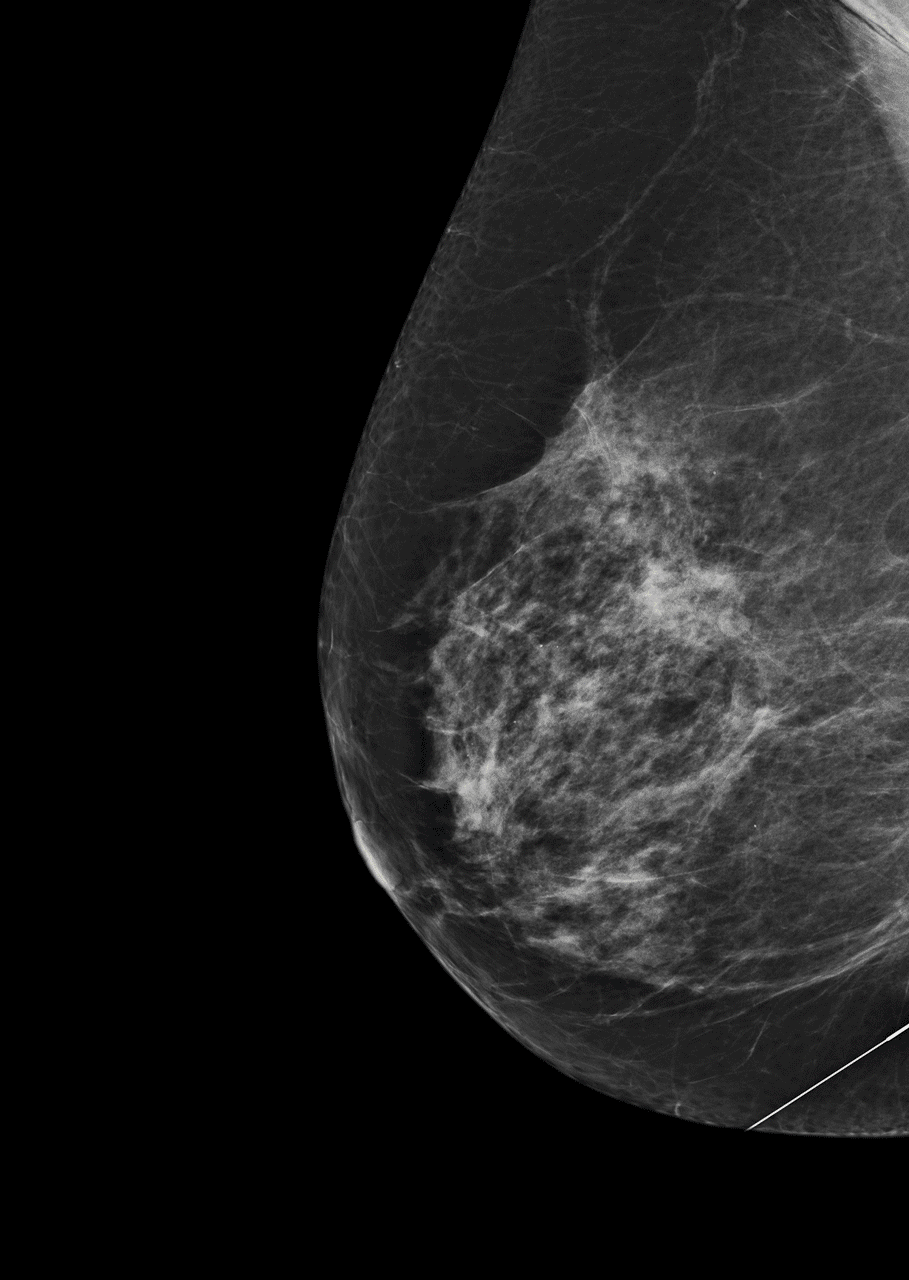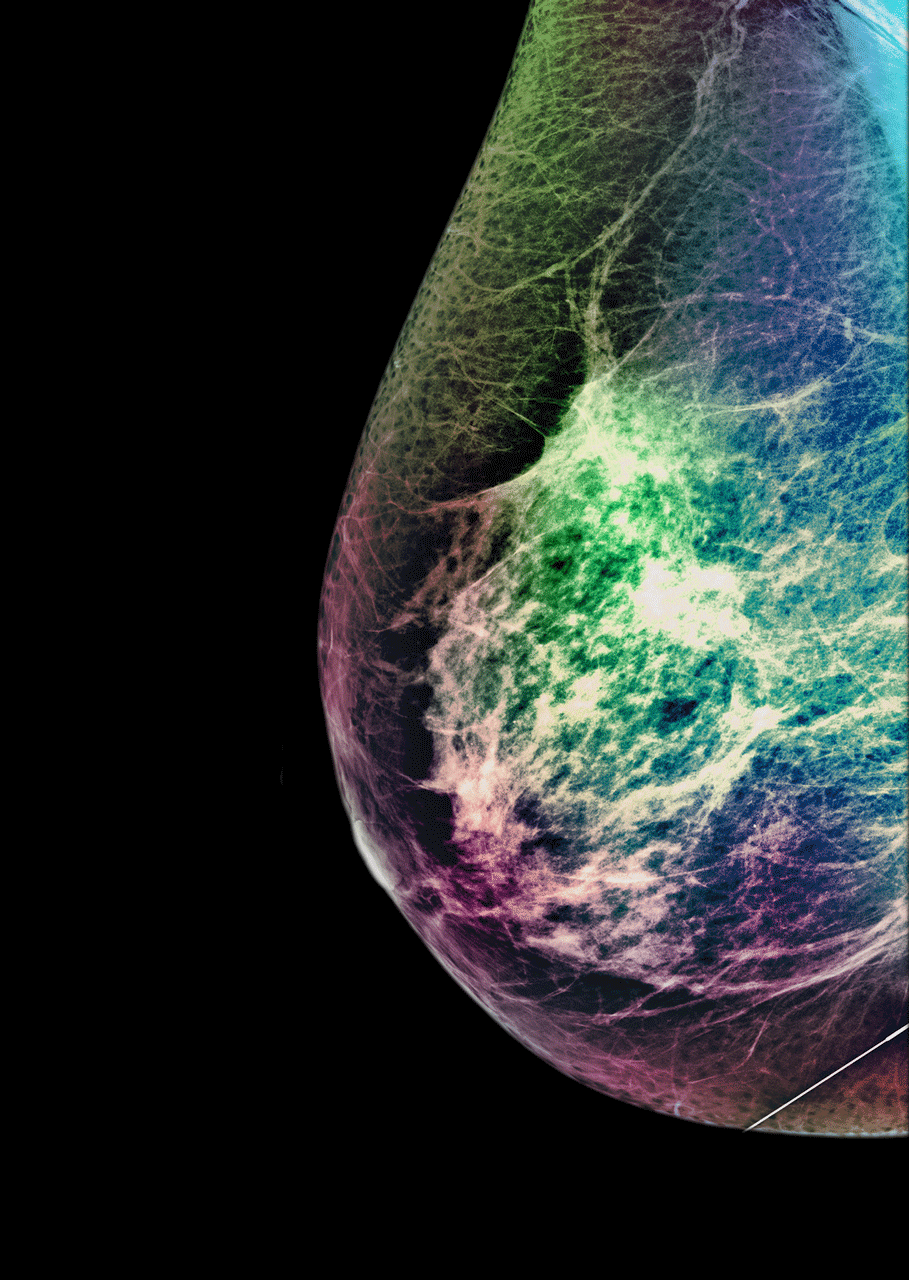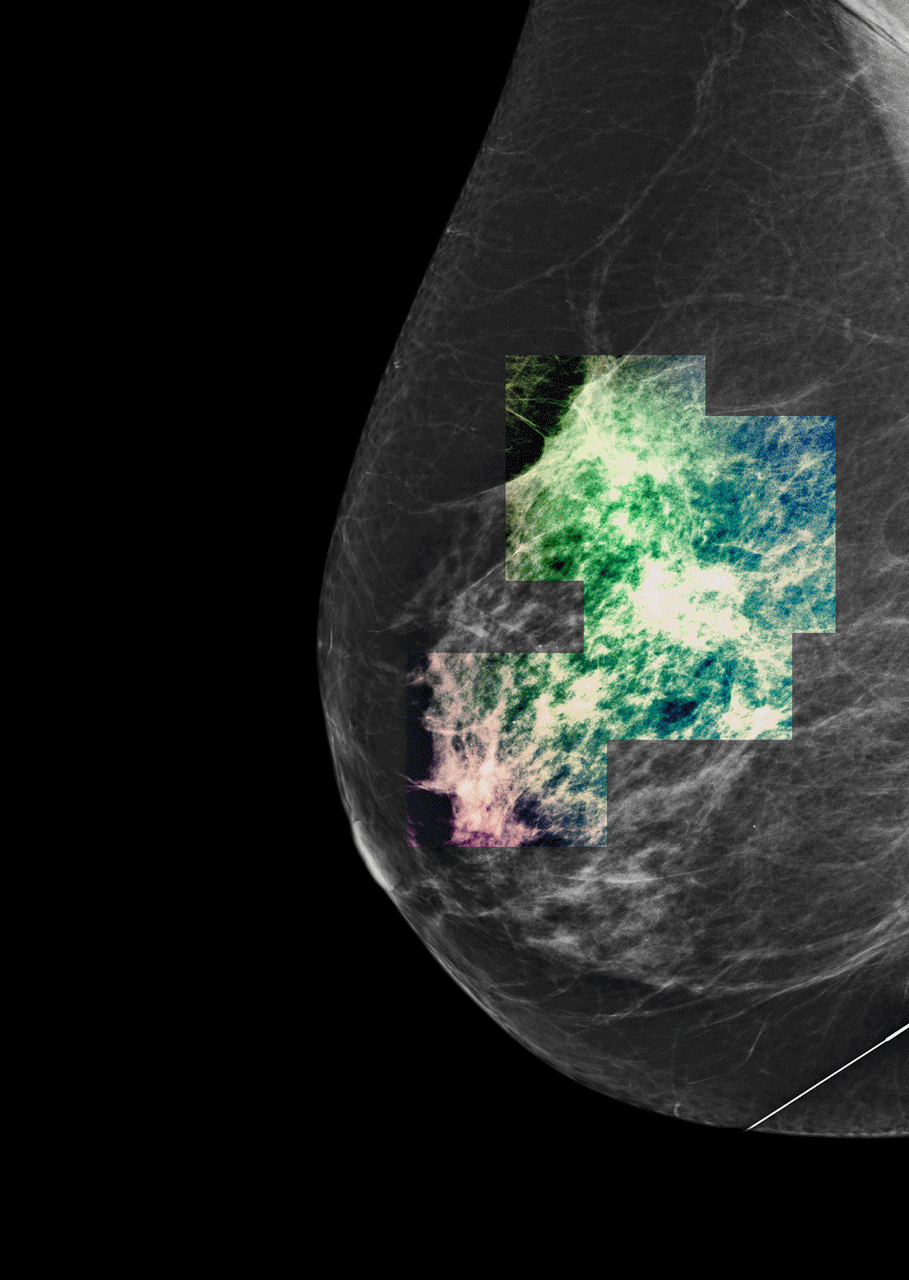
As the team started processing thousands of samples, just like algorithms in digital communication, they improved. A study of the technology found common disease-driving mutated genes detected by Guardant360 in breast, lung, colorectal, and other cancers were also present in 94-100 per cent of the solid tissues extracted from trial participants.
The test is now considered to be one of the most comprehensive on the burgeoning liquid biopsy market, looking at mutations in 70 cancer-related genes.
Guardant has processed over 15,000 liquid biopsies and 50 tumour types to improve performance and has helped doctors discover the presence of cancerous tumours before they have presented symptoms.
The technology can easily and painlessly monitor cancer, which is always evolving and changing, so treatment plans can be adapted accordingly. To monitor lung cancer, for example, a biopsy is necessary, which in the US costs $14,000 per patient and has a 19 per cent complication rate.
Once cancer signals are unlocked, at an extremely high fidelity for each type, they are classified and grouped into different sub-sets of cancer. As specificity and sensitivity gets better, the company will classify more sub-types of cancer and start decoding and unlocking signals for Stage One, Stage Two and recurrent cancers.
There is no standardised approach to the handling of quality data from genome sequencing – it differs from one hospital to another. Yet if not managed correctly, data can become corrupted, making it extremely biased or inaccurate.
In 2011, Swiss-based data medicine company Sophia Genetics set out to ‘develop an algorithmic technology that would make genomic data more accurate for diagnostics’, essentially cleaning and standardising it.
The company developed an analytics platform called Sophia DDM and an AI called SOPHiA to help hospitals overcome the bottleneck of analysing complex data generated by genome sequencing. The user in the lab loads raw encrypted data into Sophia DDM platform, the AI transforms gene data into digital information and ejects anything that could bias or make it present false results, usually a major problem in diagnosis. It then physically deciphers the molecular profile of the cancer or patient and annotates information.
“The only way you can correct the data is by asking about its production. This is the secret of SOPHiA,” says Jurgi Camblong, CEO of Sophia Genetics. “The AI has been exposed to a lot of bias so it can infer information from noise and run and trap that noise so the outcome of the data is accurate – it is not going to tell you something is there when it is not,” he says.
The company worked closely with hospitals and was routinely exposed to problems, which it worked to solve with the algorithm. The more problems it overcame, the more the algorithm learned and now the AI doesn’t need any further data to be accurate. The service is charged on a per-patient basis to make it more affordable for clinics.
Read the full article here
